One of the biggest headaches in chemistry is trying to imagine three-dimensional shapes while looking at a flat textbook. You are told that methane is a “tetrahedron,” but on paper, it looks like a flat cross.
Today we are going to solve that. We will use the Online Visualizer to apply VSEPR Theory (Valence Shell Electron Pair Repulsion) and see how atoms really organize themselves in space.
What is VSEPR Theory?
Basically, electrons hate each other. Since they have a negative charge, electron pairs forming bonds (or lone pairs) try to get as far away from each other as possible. This repulsion is what gives the molecule its shape.
To understand it, reading is not enough. You have to see it.
👉 ENTER THE ONLINE VISUALIZER HERE
(Instructions: Open the tool, tap the Magnifying Glass 🔍 and search for the molecules mentioned below).
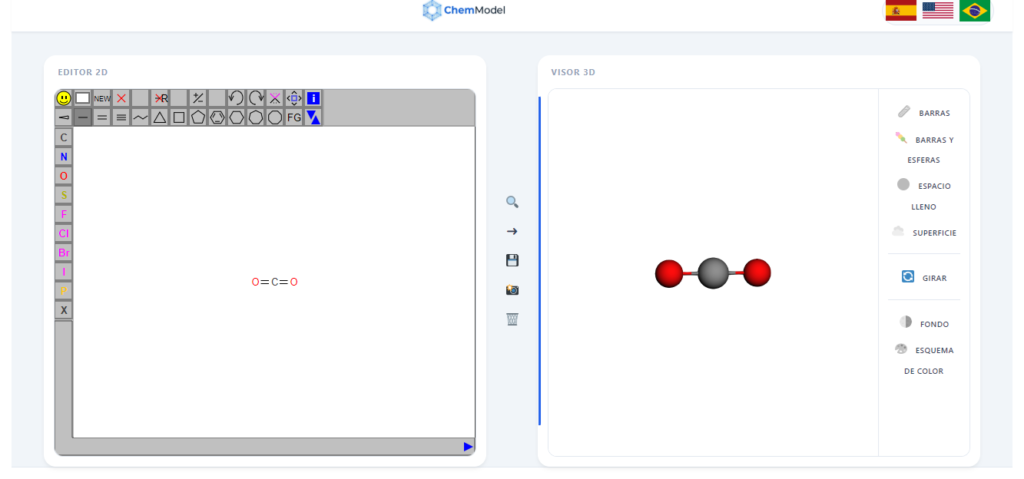
1. Linear Geometry (180∘)
The classic example is Carbon Dioxide (CO2).
- In the Visualizer: Search for “Carbon Dioxide”.
- What to observe: You will see that the three atoms are perfectly aligned. The electrons in the double bonds push against each other until they are on opposite sides.
- Recommended Style: Use the “STICKS & SPHERES” mode to clearly see the alignment.
2. Trigonal Planar Geometry (120∘)
Search for Boron Trifluoride (BF3) or draw it in the 2D editor.
- What to observe: Imagine an equilateral triangle. The Boron atom is in the center and the three Fluorines are at the corners, all on the same flat plane.
3. Tetrahedral Geometry (109.5∘)
This is where drawing on paper fails. Search for Methane (CH4).
- The paper error: In your notebook, methane looks like a 90-degree cross.
- The 3D reality: In our visualizer, you will see it is a tripod. Rotate the molecule using the “SPIN” button or your mouse. No matter how you look at it, there are always atoms pointing towards you and away from you. That is the magic of the tetrahedron.
4. The Effect of Lone Pairs: Water (H2O)
Search for “Water”. Although oxygen has 4 electron zones (like methane), two of them are lone pairs (invisible but powerful). These pairs push the hydrogens downward, closing the angle from 109.5∘ to about 104.5∘.
- Try this: Change the style to “SPACEFILL”. You will see how oxygen “occupies” a lot of space on top, squeezing the hydrogens.
Challenge for your Exam:
Use the Online Visualizer to predict and verify the geometry of these molecules:
- Ammonia (NH3): Is it flat or pyramidal?
- Sulfur Hexafluoride (SF6): An impressive octahedral structure.
Chemistry is not flat. Stop memorizing angles and start visualizing them; that is the key to mastering VSEPR. Use our tool to do your homework and understand the real structure of the universe at the atomic scale!