Have you ever wondered what actually “wakes you up” in the morning? We all know the effect of coffee, but few have seen the true protagonist up close: 1,3,7-trimethylxanthine, better known worldwide as Caffeine.
Today, we aren’t just going to review its theoretical chemical formula; we are going to manipulate, rotate, and analyze it as if you held it in the palm of your hand, using our free Online Visualizer.
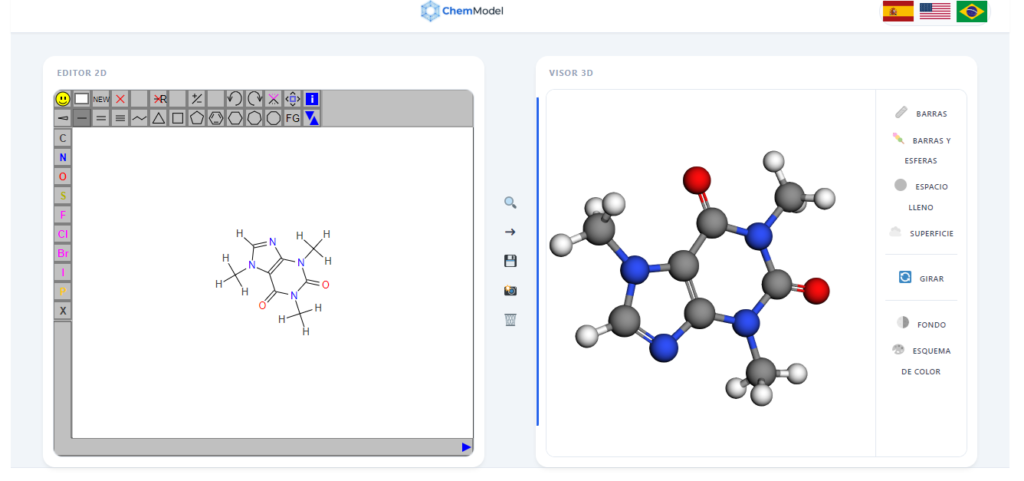
What is Caffeine chemically?
Caffeine is an alkaloid of the xanthine group that acts as a central nervous system stimulant. For chemistry students, it is a fascinating molecule because, despite being small, it contains a wide variety of functional groups and key structural characteristics.
Technical Data:
- IUPAC Name: 1,3,7-trimethylpurine-2,6-dione
- Chemical Formula: $C_8H_{10}N_4O_2$
- Molar Mass: 194.19 g/mol
- Hybridization: Most of its carbon and nitrogen atoms exhibit $sp^2$ hybridization, giving it a mostly planar structure.
Use the Online Visualizer Now
Forget about installing heavy software. We have prepared an interactive simulation for you to study this molecule directly in your browser.
👉 CLICK HERE TO OPEN THE ONLINE VISUALIZER
(Quick Instructions: Upon entering, tap the Magnifying Glass 🔍 in the center bar and type “Caffeine” to load the model instantly).
Structural Analysis: What should I look for?
Once you have the molecule loaded in the Online Visualizer, we recommend using the “Styles” tool (Right Bar) to identify the following parts:
- The Ring System:Caffeine is a heterocyclic molecule. You will notice it consists of two fused rings: a six-membered ring (pyrimidine) and a five-membered ring (imidazole). This fused structure is known as Purine.
- The Methyl Groups ($CH_3$):If you change the style to “Spacefill”, you will see three bulky groups sticking out. These are carbons bonded to three hydrogens (methyl groups) located at positions 1, 3, and 7 of the rings.
- Color Code (CPK):In our visualizer, atoms follow the international standard:
- Gray: Carbon ($C$)
- White: Hydrogen ($H$)
- Blue: Nitrogen ($N$) – Notice the 4 key atoms within the rings.
- Red: Oxygen ($O$) – Observe the characteristic double bonds of the ketones.
The Biological Connection: Caffeine vs. Adenosine
Here is the secret to why you don’t feel sleepy. If you use the visualizer’s 2D Editor to draw the Adenosine molecule, you will notice something surprising: its base structure is incredibly similar to that of Caffeine.
Due to this resemblance, caffeine acts as an “imposter.” It binds to adenosine receptors in your brain but does not activate them. It simply blocks them, preventing real adenosine (which is what signals fatigue) from doing its job.
Practical Task for Students:
To master the use of the Online Visualizer, try the following:
- Enter the tool on the homepage.
- Search for “Caffeine”.
- Change the viewing mode to “Surface”. This will allow you to see the actual volume the molecule occupies in space (Van der Waals Radius).
- Use the “PNG Capture” 📸 button in the center bar to download a clean image and use it in your presentation or school assignment.
Chemistry is much easier to understand when you can see it in three dimensions!